Vaccine Not FDA-Approved for Kids Under Five
Government regulators want more information before approving the Pfizer and BioNTech Vaccine for those under five.
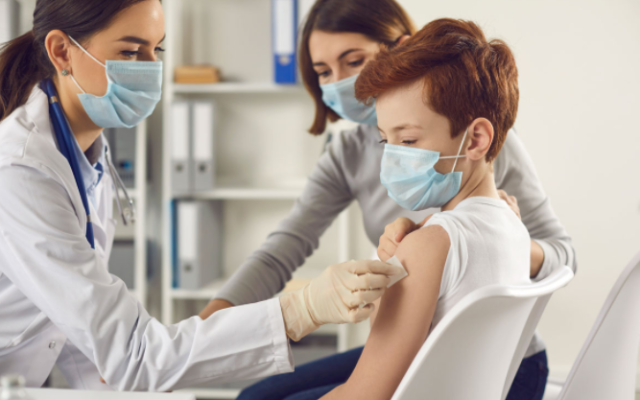
Parents will have to wait until at least early in April to learn whether their children under five will need three shots to be protected against COVID-19.
A delay in approving the vaccine from Pfizer and BioNTech for young children came Feb. 11 after the FDA said they needed more data. That is when the two companies pulled their application for approval to vaccinate children under five from the Food and Drug Administration.
Many had expected the FDA to approve the company’s two-shot dosage, even though a study of children ages two through four released in December, showed that the vaccine produced only 60 percent of the antibodies as compared to levels in older teens and young adults.

In a prepared statement, the pharmaceutical manufacturers maintained that a third shot may be needed for the 18 million children in America under five.
According to a spokesman for the manufacturer, “Given that the study is advancing at a rapid pace, the companies will wait for the three-dose data as Pfizer and BioNTech continues to believe it may provide a higher level of effectiveness in this age group.”
Since the vaccine was approved for children ages five to 11 nearly four months ago federal government statistics show only about 50,000 to 75,000 doses are being administered each day. According to a study in January 2022 by the Kaiser Family Foundation, only 18.8 percent of that age group are fully vaccinated and only 28.1 percent have received at least one dose.
In Georgia, where on Feb. 14 Governor Brian Kemp announced plans to support legislation to give parents, not educators, the right to decide if children must wear masks in school, those numbers were much lower. According to federal data of the state of Georgia, the Kaiser study showed only 9.2 percent had two shots and only 16.7 percent had one shot. In the neighboring state of Alabama only 5.3 percent of children in the age group were fully vaccinated.

According to Dr. Howard Silverman at Roswell Pediatric Center, the group practice of 16 physicians, has given out about 4,000 COVID shots.
He says that the parents of most of the children in the five to 11 age group prefer to consult a physician for counseling rather than getting the shots at a local pharmacy.
He points out that he frequently must explain the benefits of the shots for those who are hesitant to agree to have their children receive them. “There has been resistance for the older kids, and the younger kids. You get those who are very interested, but you also get those who say, ‘you are not doing that to my child,’ and there are those who want to wait a month or two. It’s an attitude that really doesn’t change whether you are talking about the 16 and older age group, the 12- to 16-years-olds or those five- to 11-years-old. It has been the same case all the way around.”
Some health observers fear interest in the vaccine may wane even further as the number of serious cases in children continues to drop. Silverman says that February has been far better than the end of last year and January of this year.
Silverman added, “Things have improved dramatically. The Omicron variant looks like it has been mild in pediatric patients. We have admitted a few young patients to the hospital with respiratory conditions, usually for a couple of days and they’ve gone home. But the overwhelming number of patients that we’ve seen, and we’ve seen a lot of patients with COVID, have had coughs, congestion, sore throat, fever just those kinds of symptoms.”
Even though some children have developed COVID without apparent symptoms, studies have shown that they have the potential to spread the virus as effectively as adults.
Because of that Dr. Evan Anderson, a pediatrician who runs the Vaccine Research Center for Children’s Healthcare of Atlanta was among those who was an early proponent of COVID vaccines for children. Not just for the protection it gives them but for how they may help to dampen the spread of the virus.
He told a medical writer for Emory University this month that children are a prime source of infection in the home. He said, “Among the most important methods of COVID-19 transmission are from inside the household and from household visitors. Having a vaccine available for our children will have the potential to significantly impact transmission to parents, grandparents and other adults.”
Today’s dangerously high drop in child COVID vaccinations is in sharp contrast to a time just ten months ago when Anderson was beginning the first clinical trials of the vaccine on five- to 11-year-olds. In May 2021 when he called for 400 children as volunteers for the research, there were 2,000 applicants.
With questions about the effectiveness of the vaccine among preschoolers still lingering, and the possibility that it may require three shots instead of two to protect them, parents could continue to resist having their children vaccinated. Which may mean that efforts to add those under five years old to those Americans who are protected against the infection this spring, may face significant new challenges.
- Education
- camp
- Local
- Bob Bahr
- COVID
- COVID-19
- pandemic
- Pfizer
- BioNTech
- FDA
- US Food and Drug Administration (FDA)
- Vaccine
- children
- Kaiser Family Foundation
- Governor Brian Kemp
- Dr. Howard Silverman
- Roswell Pediatric Center
- omicron
- Omicron variant
- Dr. Evan Anderson
- Vaccine Research Cente
- Children’s Healthcare of Atlanta at Egleston
- Children's Healthcare of Atlanta
- Emory University



comments